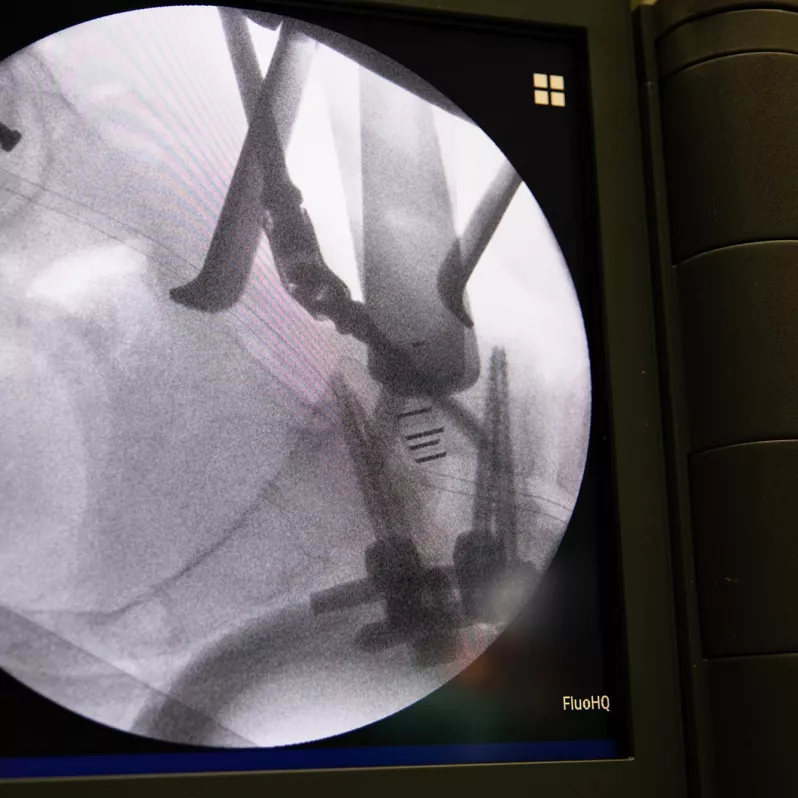
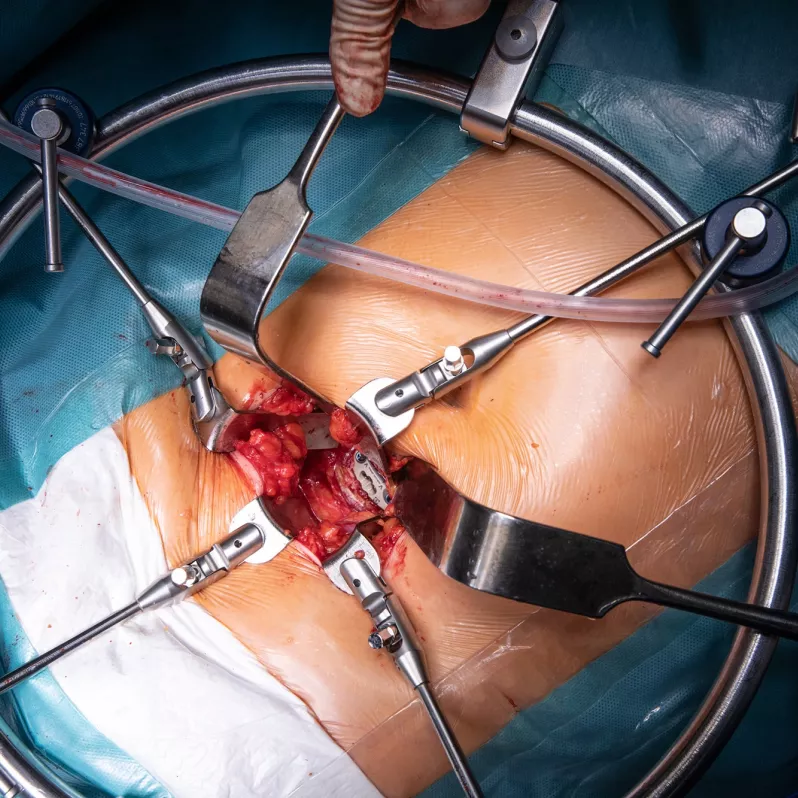
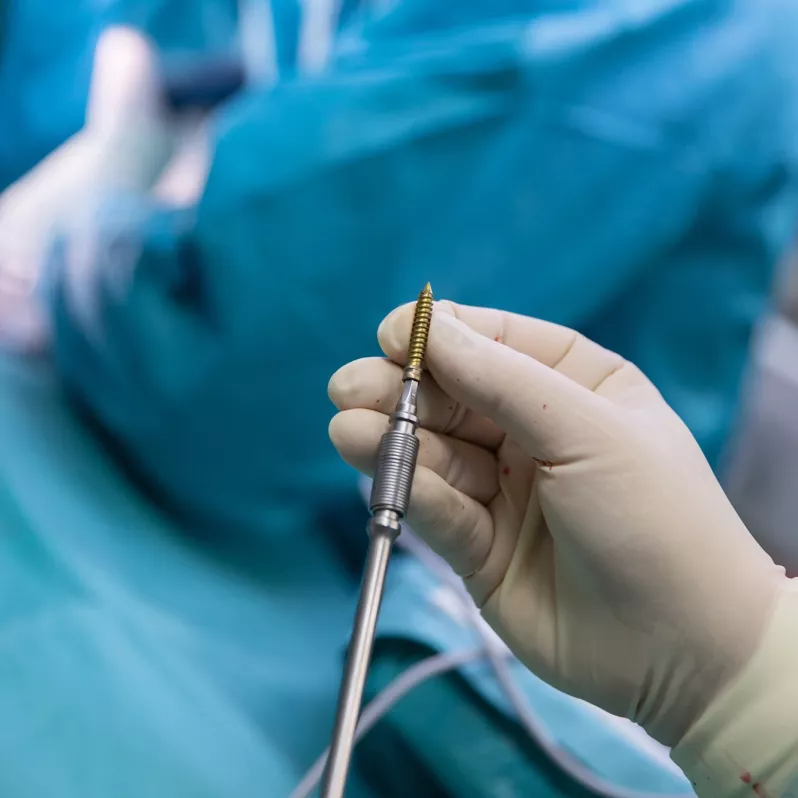
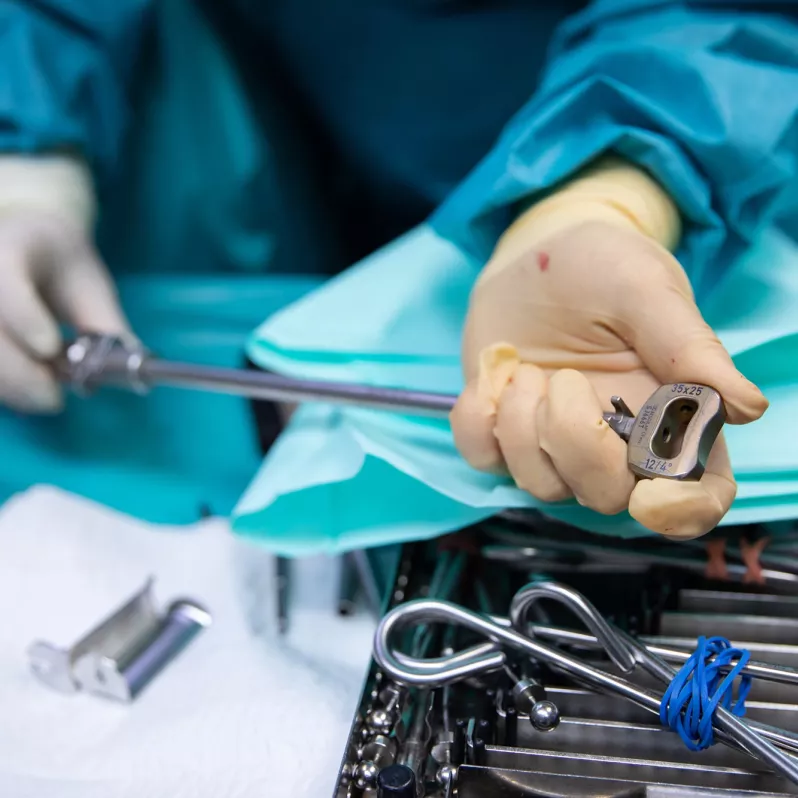
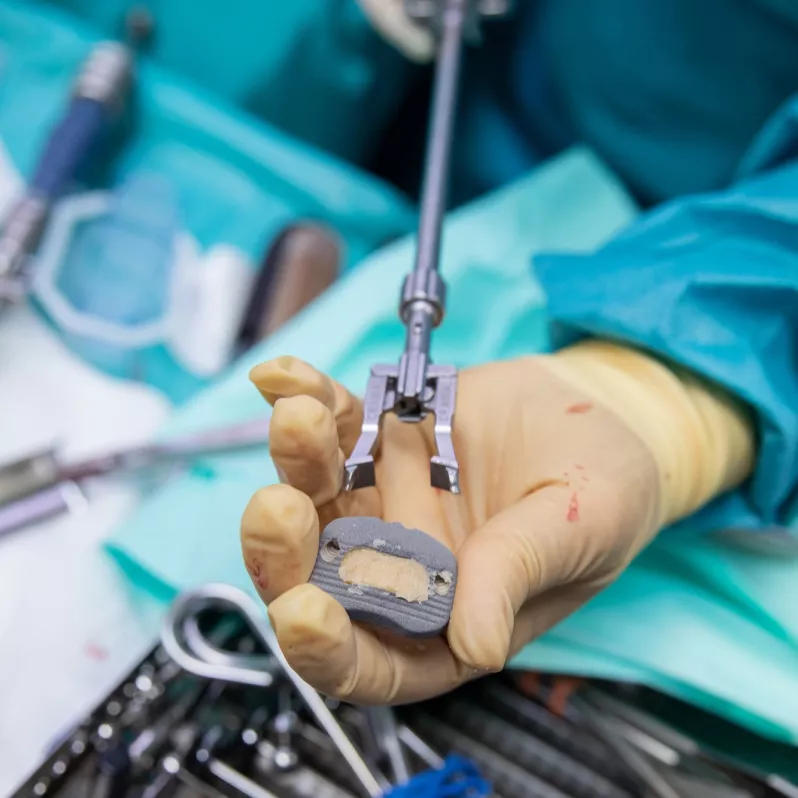
Arcadius XP®
The stand-alone interbody fusion system produced by Aesculap AG used to stabilize the lumbar spine in L2-S1 segments through an anterior approach.
Who is it for?
Interbody fusion system Arcadius XP® is indicated for patients with degenerative disorders and instability of the intervertebral disc (DDD), spondylolisthesis up to grade 1 and those suffering from post-discectomy syndrome or post-traumatic instability. With four diverging bone screws it is capable of self-fixation without the use of an additional anchorage system.
Why Arcadius XP®?
The Arcadius XP® implant combines stability, high quality imaging and elegance of design. Its surface is covered with a porous biocompatible PlasmaporeXP® coating made of titanium, which guarantees increased osteoconductivity and initial stability in contact with the bone surface. This is also ensured by four self-tapping bone screws with a divergent design that can be easily accessed from all angles. The integrated double locking mechanism with one-step activation further contributes to stability and efficient workflow during the actual surgical procedure.
Good intraoperative and postoperative visibility of the Arcadius XP® system is ensured by five tantalum markers and the PlasmaporeXP® surface itself. Thanks to the wide range of sizes available, the implant can be customised to the specific anatomical requirements of each patient.