obelisc® + MACS II
obelisc®
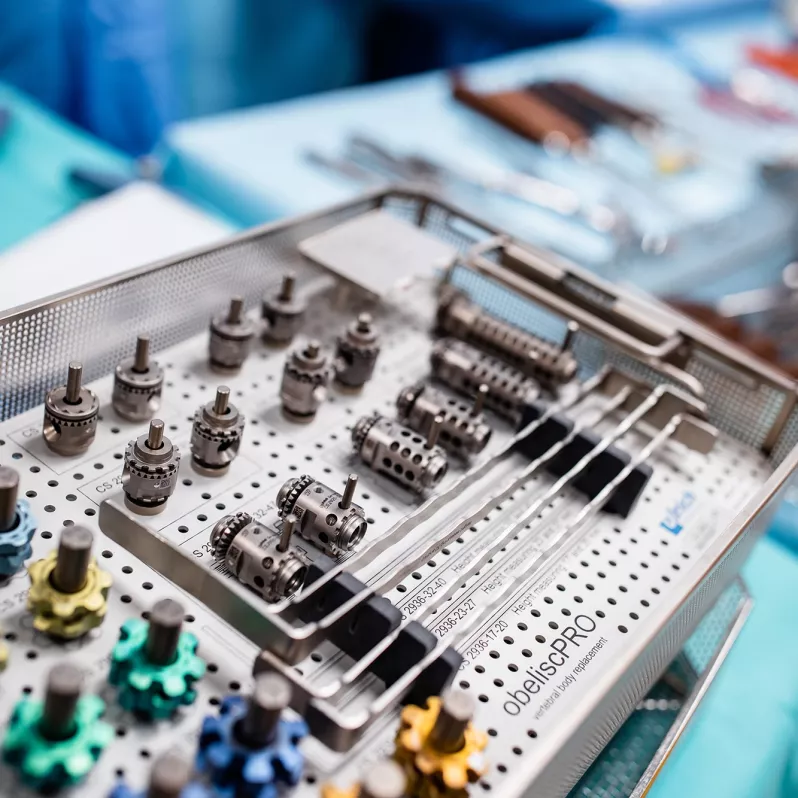
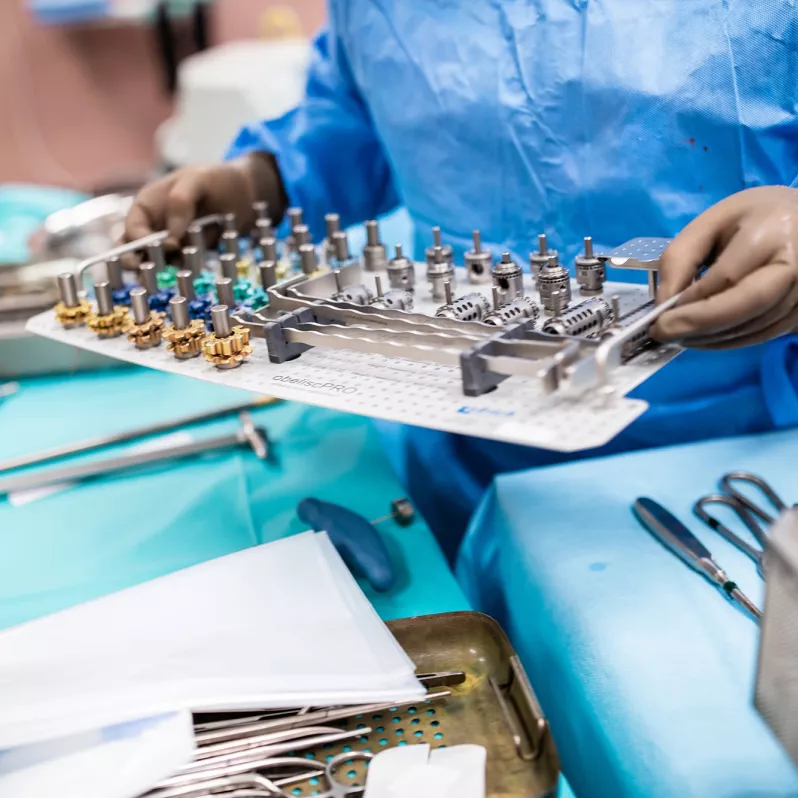
The vertebral body replacement obelisc® produced by German company ulrich medical is used for surgical reconstruction of substance defects of the anterior thoracic and lumbar spine.
Who is it for?
The implant is indicated for patients who have undergone a complete or incomplete corpectomy due to destruction of a vertebral body by e.g. tumor, fracture or inflammation. It allows to bridge the resulting defect up to a height of more than 130 mm, which in practice means replacing one or more spinal segments. The obelisc® implant is intended to be used in combination with another stabilisation system, such as the pedicle screw-bar system.
Why obelisc®?
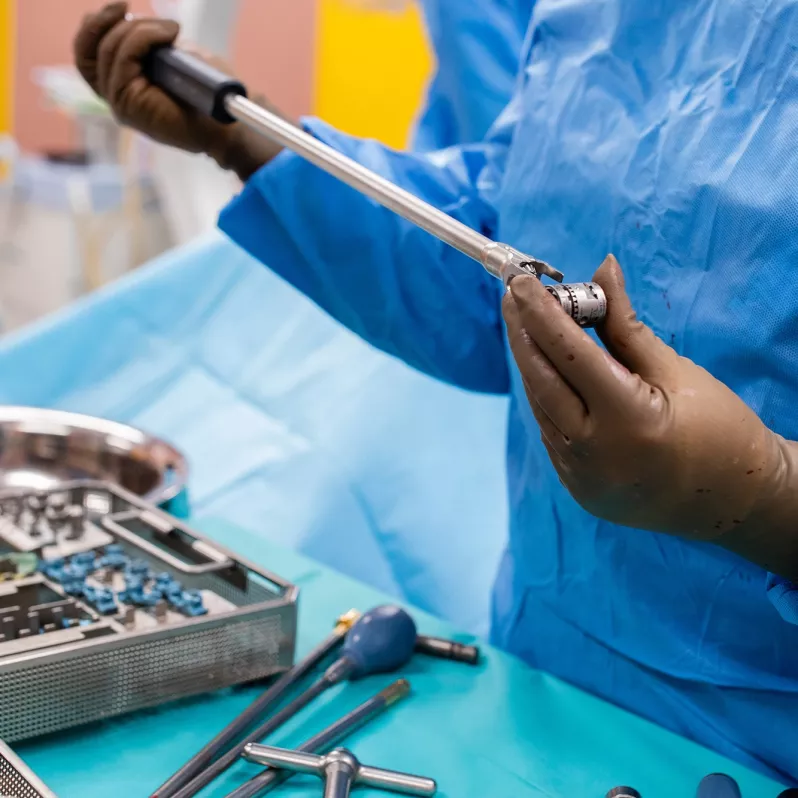
Thanks to its innovative construction the vertebral body replacement allows for implantation of a minimally invasive nature and expansion directly in situ. The bevel gear drive in the loading device is designed to precisely adjust the required expansion, which can vary from approximately 20 to 130 mm depending on the nature of the procedure. The final fixation is carried out by means of a locking screw, which ensures high postoperative stability.
This stability is also contributed to by a variable system of end pieces, which easily adapt to the individual physical requirements of each patient. The serrations on these end pieces serve to anchor them even better to the vertebra cover plates. The implant itself is designed solely to capture axial compression forces.
MACS II
The modular construct system produced by German company Aesculap AG is used for anterior stabilization of the thoracic and lumbar spine.
Who is it for?
This system is suitable for patients suffering from degenerative disorders of the intervertebral discs due to spondylolisthesis, spondylolysis or spinal stenosis. It is also intended for stabilisation of post-traumatic instabilities and deformities or spinal tumors. Its design allows correction and fixation of one or more segments simultaneously from an anterolateral approach.
Why MACS II?
The MACS II system represents the next step in the development of stabilisation implants and is a direct successor to the previous biomechanically well-proven MACS PL® system. A fundamental change is the overall simplification of the tools and operating procedure using new pre-mounted set screw with centralizer guides. All parts of the implant are made of titanium alloy, except for the pre-mounted set screw which is made of pure titanium.
The MACS II provides its recipients with high stability combined with safety thanks to a firmly locking mechanism and the use of monocortical screws. Thanks to its modular concept, it meets the needs of a wide range of patients while allowing surgeons to use less invasive techniques. its design contributes to the smooth integration of the implant into the spinal structure. Possible postoperative manipulation is also possible through a microsurgical or endoscopic approach.