uCentum® + TSpace XP®
uCentum®
The uCentum® system from the German company ulrich medical is a comprehensive implant system for posterior stabilization, fixation and correction of the thoracic and lumbar spine, including the sacrum.
Who is it for?
Implant recipients may suffer from degenerative and post-traumatic spinal instability, disc disease, scoliosis, spinal stenosis, spinal fractures or deformities, pseudoarthrosis, spondylolisthesis, spinal tumours or post-discectomy syndrome. The system is intended for open or minimally invasive percutaneous applications.
Why uCentum®?
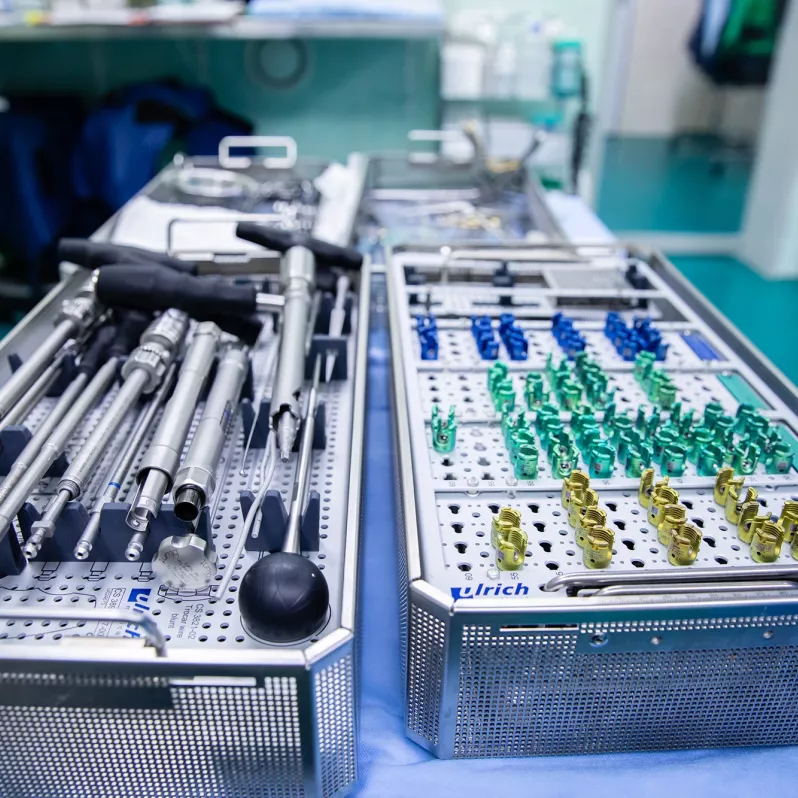
The uCentum® system is a top-quality, self-contained solution for a wide range of thoracic and lumbar spine procedures, even in minimally invasive procedures. It is based on a set of cannulated and not cannulated pedicle screws. These augmentable screws allow for cement augmentation of the vertebral body in osteoporotic spinal disorders. Furthermore the crosslink and offset connectors, lamina hooks and rods made of cobalt-chromium alloy allow for more effective correction of spinal deformities and greater postoperative stability than its titanium equivalents.
Components are supplied in a range of anatomical sizes to meet the needs of as many patients as possible. In addition, the uCentum® system is suitable for right-to-left instrumentation, making it easier to adapt to the individual needs of a left-handed surgeon.
TSpace XP®
The transforaminal lumbar interbody fusion system by the German company Aesculap AG is used for the stabilazation of single or multiple segments of the lumbar spine.
Who is it for?
TSpace XP® system is designed for patients suffering from degenerative and post-traumatic instabilities of the spine, spondylolisthesis or postdiscectomy syndrome. Thanks to its construction, it allows implantation through an open or minimally invasive approach. It should be used in combination with an internal fixator, for example the Ennovate® system, at all times. For functional reasons, this system is not advisable when injury or disease has resulted in disruption of the bone structures of the spine that would interfere with stable anchorage of the implant.
Why TSpace XP®?
The implant consists of a combination of intelligent, next-generation biocompatible materials. PEEK OPTIMA® provides the core with sufficient mechanical strength and fatigue resistance while maintaining excellent imaging properties for intraoperative and postoperative X-rays and CT scans. Titanium PlasmaporeXP® coating boasts proven osteoconductive properties and porosity of up to 60 % which creates an optimal surface-to-bone contact for bone ingrowth leading to further stabilization.
The entire TSpace XP® system is designed for easy implantation into a damaged disc and provides maximum security against subsequent migration, which is taken care of by the increased roughness of the surface in addition to a serrated profile. To ensure anatomical compatibility with the patient’s body, the implant is available in a range of sizes.